Self Cultivation to develop internal chi and internal power that eliminates self doubt, anxiety, grief, disappointment and live in a state of grace. Free from hate, fear, malice, pride, jealousy, worry, attachment, and self-centeredness.
Thursday, November 13, 2014
Tuesday, November 11, 2014
Grow Rich with Diamonds
Diamonds have two great economic properties. Portability and appreciation over inflation. It's not too late to buy and invest in diamonds.
There is nothing in the word that compares to diamond investment as a physical asset. For over the years diamonds have steadily increased in value at a rate that exceeds inflation. It is estimated that diamond appreciate at a rate of 12 percent a year despite wars, recessions, depressions, stock market crashes, etc.
Diamonds are the safest investment that you own. Diamonds are financially secure. Diamonds are the only one investment that behave predictably.
Diamond banking.
Diamonds will always be a safe, profitable investment asset.
Diamonds have been a part of history as a collectible and as an investment.
Diamonds will always be a valuable asset/commodity and a possible alternative investment class.
Diamonds are the most viable investment option. They are the ultimate investment class. They are so ahead of anything else, that the difference is astonishing to anyone who understands this.
Diamonds are inflation positive.
Unlike paper money, diamonds can not be printed indiscriminately. Diamonds need to hard labor to mine. Skill to cut. Certification to be obtained.
Unlike gold it is not subject to major upswing. It can not be debased with other metals. Large quantities of diamonds are portable unlike gold.
Unlike stocks, diamonds are inflation positive after taxes. Unlike real estate diamonds are portable and liquid. Unlike other gemstones, diamonds have a track record of trust and confidence. Unlike food and technology, diamonds do not do obsolete.
Diamonds can grant you the power to survive. In financial panic, diamonds can help you survive and prosper.
Diamonds are the oldest survival money in the world. Because they provide a means to survive, diamonds are in reality a financial time machine. They have the power to carry the owner in a time of peril. With a world in economic chaos, diamonds have the power to transport our wealth and ourselves into another city, state or country.
Currency speculative
Real Estate has high entry costs
Stocks and Mutual Funds (No control over the price/market).
The problems with diamonds is capital loss, depreciation, it does not pay interest, it needs marketing and man agent attention, it can be stolen.
There is nothing in the word that compares to diamond investment as a physical asset. For over the years diamonds have steadily increased in value at a rate that exceeds inflation. It is estimated that diamond appreciate at a rate of 12 percent a year despite wars, recessions, depressions, stock market crashes, etc.
Diamonds are the safest investment that you own. Diamonds are financially secure. Diamonds are the only one investment that behave predictably.
Diamond banking.
Diamonds will always be a safe, profitable investment asset.
Diamonds have been a part of history as a collectible and as an investment.
Diamonds will always be a valuable asset/commodity and a possible alternative investment class.
Diamonds are the most viable investment option. They are the ultimate investment class. They are so ahead of anything else, that the difference is astonishing to anyone who understands this.
Diamonds are inflation positive.
Unlike paper money, diamonds can not be printed indiscriminately. Diamonds need to hard labor to mine. Skill to cut. Certification to be obtained.
Unlike gold it is not subject to major upswing. It can not be debased with other metals. Large quantities of diamonds are portable unlike gold.
Unlike stocks, diamonds are inflation positive after taxes. Unlike real estate diamonds are portable and liquid. Unlike other gemstones, diamonds have a track record of trust and confidence. Unlike food and technology, diamonds do not do obsolete.
Diamonds can grant you the power to survive. In financial panic, diamonds can help you survive and prosper.
Diamonds are the oldest survival money in the world. Because they provide a means to survive, diamonds are in reality a financial time machine. They have the power to carry the owner in a time of peril. With a world in economic chaos, diamonds have the power to transport our wealth and ourselves into another city, state or country.
Currency speculative
Real Estate has high entry costs
Stocks and Mutual Funds (No control over the price/market).
The problems with diamonds is capital loss, depreciation, it does not pay interest, it needs marketing and man agent attention, it can be stolen.
Monday, November 10, 2014
Health is the paramount of all success
Health is the Paramount of all Success is Health and Happiness:
Healthy Mind
Healthy Emotion
Healthy Spirit
Healthy Organs
Healthy Body
No Stress
No Speculation
The future is in your hands.
Healthy Mind
Healthy Emotion
Healthy Spirit
Healthy Organs
Healthy Body
No Stress
No Speculation
The future is in your hands.
Sunday, November 09, 2014
Diamond as an Investment
Are diamonds a good investment? Are diamonds are rare? What are the advantages and disadvantages of investing in diamonds? What should one look for when investing in diamonds? What are the difference when investing in diamonds compared to stocks, real estate gold, art?
How does it correlate to real estate and stocks?
There is a positive correlation between diamonds and other assets such as stocks and real estate.
Why do people invest in diamonds? Why do people buy diamonds?
Diamonds are heterogeneous in nature which means that every diamond is unique. No two diamonds are the same. I would consider gold homogenous in nature.
What is a diamond?
A diamond is a highly desirable dispersive "precious" mineral.
What are the advantages when investing in diamonds?
1. It is perceived to have high intrinsic value
2. It is tangible asset that can be converted into cash
Mobility (Portability)
What are the disadvantage of investing in diamonds?
1. It bears no interest
2. It is somewhat illiquid
3. Diamonds are not fungible as each diamond is unique (Gold is fungible cause an ounce of gold is an ounce of gold).
How does it correlate to real estate and stocks?
There is a positive correlation between diamonds and other assets such as stocks and real estate.
Why do people invest in diamonds? Why do people buy diamonds?
Diamonds are heterogeneous in nature which means that every diamond is unique. No two diamonds are the same. I would consider gold homogenous in nature.
What is a diamond?
A diamond is a highly desirable dispersive "precious" mineral.
What are the advantages when investing in diamonds?
1. It is perceived to have high intrinsic value
2. It is tangible asset that can be converted into cash
Mobility (Portability)
What are the disadvantage of investing in diamonds?
1. It bears no interest
2. It is somewhat illiquid
3. Diamonds are not fungible as each diamond is unique (Gold is fungible cause an ounce of gold is an ounce of gold).
Bhagat Singh Thind
If you feel doubtful in someones presence, take my advice and keep silent.
How far two or more minds can get along in mutuality depends on the Degree of their sincerity towards others. The common ground has to be made, and kept, by moral effort.
Conquer the Worlds of Ups and Downs
Sow today what you would love to harvest in the days to come .
If you think you’re green you grow, if you think you’re ripe you rot.
Nothing fails like success.
Get all things, but be possessed by none.
If your nature is crooked, purify it. First exercise self-control. Control the nature.
How far two or more minds can get along in mutuality depends on the Degree of their sincerity towards others. The common ground has to be made, and kept, by moral effort.
Conquer the Worlds of Ups and Downs
Sow today what you would love to harvest in the days to come .
If you think you’re green you grow, if you think you’re ripe you rot.
Nothing fails like success.
Get all things, but be possessed by none.
If your nature is crooked, purify it. First exercise self-control. Control the nature.
Tuesday, September 02, 2014
Warren Cassell Jr.
I have a dedication of achieving great success.
"Study nature, love nature, stay close to nature. It will never fail you." Frank Lloyd Wright
"It occurred to me that nature is constantly trying to teach us something."
Nature is the source of unlimited knowledge.
I became highly knowledgeable about plants and the many different functions and features of nature.
I observed the movements of every creature in my presence.
The lessons that are taught in self-help seminars and even investing principles are all aligned with what I observed in nature and the lives of other animals.
When we adopt the lifestyle of nature we position ourselves closer to achieving success--wealth, health and happiness!
When we try to harm others we set ourselves up to be harmed as well. When a bee harms someone, it is the bee that dies.
The more in tune we are with nature and the more we actually observe our surroundings, the easier it is for us to adapt to change which results in more effective evolution.
Do not be afraid to surface every now and then. It helps lessen the distractions of your regular life, leaving a lot of space in your mind for new thoughts to flow in and out.
Nature holds an abundance of success principles.
Nature can teach us how to succeed in business and in life.
Keep moving forward!
Pausing is a form of moving forward.
It is vital that you try something new.
You will never be able to maximize the talent of your staff if you never take the risk to invest in new ventures.
Change can be caused by a shift in the demand of what consumers want.
Change causes pain but we should not resist change.
"Study nature, love nature, stay close to nature. It will never fail you." Frank Lloyd Wright
"It occurred to me that nature is constantly trying to teach us something."
Nature is the source of unlimited knowledge.
I became highly knowledgeable about plants and the many different functions and features of nature.
I observed the movements of every creature in my presence.
The lessons that are taught in self-help seminars and even investing principles are all aligned with what I observed in nature and the lives of other animals.
When we adopt the lifestyle of nature we position ourselves closer to achieving success--wealth, health and happiness!
When we try to harm others we set ourselves up to be harmed as well. When a bee harms someone, it is the bee that dies.
The more in tune we are with nature and the more we actually observe our surroundings, the easier it is for us to adapt to change which results in more effective evolution.
Do not be afraid to surface every now and then. It helps lessen the distractions of your regular life, leaving a lot of space in your mind for new thoughts to flow in and out.
Nature holds an abundance of success principles.
Nature can teach us how to succeed in business and in life.
Keep moving forward!
Pausing is a form of moving forward.
It is vital that you try something new.
You will never be able to maximize the talent of your staff if you never take the risk to invest in new ventures.
Change can be caused by a shift in the demand of what consumers want.
Change causes pain but we should not resist change.
Friday, August 22, 2014
Principle of Wealth Creation
Avoid Catastrophic Risks
Experience is wealth
Labor is wealth
Sample items
Income generating assets
Network
Technologies
Stay Healthy
Keep Expenses Low
Social Capital
Avoid Debt
Stay Liquid
Frugal
Think Long Term
Get a job that pays
Maintain High Credit
Acquire Skills
Know how to Sell
Balanced Diversified Desirable Portfolio
Turning $1 Million into $18 Million in 50 years.
Compounding Wealth
Low Risk Leveraging
Experience is wealth
Labor is wealth
Sample items
Income generating assets
Network
Technologies
Stay Healthy
Keep Expenses Low
Social Capital
Avoid Debt
Stay Liquid
Frugal
Think Long Term
Get a job that pays
Maintain High Credit
Acquire Skills
Know how to Sell
Balanced Diversified Desirable Portfolio
Turning $1 Million into $18 Million in 50 years.
Compounding Wealth
Low Risk Leveraging
Monday, August 11, 2014
Principles of Diamond Investing
Treat each diamond like a business.
Buy a diamond so good that a dummy can sell it.
Own rare valuable stones will continue to produce satisfactory returns.
Certified diamond with global power.
Stay away from stones that undesirable stone.
Buy diamond priced below the market value
Buy a diamond that can be sold at a higher price.
Buy a diamond within your circle of competence
Buy a diamond within your market reach
Do not compete too heavily. Competition is hazardous to human wealth and health.
Buy a diamond so good that a dummy can sell it.
Own rare valuable stones will continue to produce satisfactory returns.
Certified diamond with global power.
Stay away from stones that undesirable stone.
Buy diamond priced below the market value
Buy a diamond that can be sold at a higher price.
Buy a diamond within your circle of competence
Buy a diamond within your market reach
Do not compete too heavily. Competition is hazardous to human wealth and health.
Sunday, June 15, 2014
Crystallized Carbon
Diamonds are essentially composed of a single element of carbon with most of them containing a bit of impurities. Most of the impurities is nitrogen.
All known forms of life are based on organic constructions of carbon. Crystalline carbon exists in alternative forms bonded in different structural arrangement called allotropes or polymorphs.
All known forms of life are based on organic constructions of carbon. Crystalline carbon exists in alternative forms bonded in different structural arrangement called allotropes or polymorphs.
Friday, June 13, 2014
Economic Geology
Economic Geology is the study of materials that are extracted from the Earth for our use in industry. The study of minerals is called mineralogy. The study of gemstones is a new study called gemology.
The earth is in continuous state of change.
The earth is in continuous state of change.
Importance of Cut
Rough gem is an unfashioned material worth making into a gemstone or ornament. Fashioning of the gem reveals their color and optical effects to the greatest degree. It also enhances their value.
What is a Gem
A gem is a rare and desirable product of our planet and diamonds are the most desired gem of all. Gem material is an material that is used for adornment, decoration, investment, symbolism, luxurious and conspicuous consumption.
A wide range of substances can be thought of as a gem. A diamond crystal, a faceted amethyst, a marble slab, a block of plastic imitating jet, a cultured pearl, a composite of gemstone glass, and garnet might all be described as gem material.
A wide range of substances can be thought of as a gem. A diamond crystal, a faceted amethyst, a marble slab, a block of plastic imitating jet, a cultured pearl, a composite of gemstone glass, and garnet might all be described as gem material.
Monday, May 26, 2014
Diamonds Classification and their impurities
Pure diamonds are composed entirely of carbon. However, diamond crystals contain minute quantities of trace elements that can take the place of some carbon atoms and the most common form is nitrogen.
A trace element is a an element that occur in minute quantities. Because of their impurities, diamonds are classified into two types. Type I and Type II.
Type I - Nitrogen occur as the major impurity up to 0.2%.
Type II - Do not contain nitrogen as a significant impurity. Type II diamonds also contain nitrogen but does not affect the physical properties.
Diamonds with large concentration of nitrogen impurities start with a single isolated atom. Due to the geological condition of heat and pressure over time, the nitrogen migrate thru the structure to form pairs and then aggregates of there or four nitrogen atoms which are Ia.
Type Ib transform into Type Ia.
The majority of natural diamonds are Type Ia.
Type Ia diamonds are diamonds that contain nitrogen atoms as pairs or small groups called aggregates.
Type IaA- diamonds contain aggregates of 2 nitrogen atoms
Type IaB - or groups of 3 or 4 nitrogens called aggregates with vacancies. Groups of 3 surrounding a vacancy are called N3 color centers.
A trace element is a an element that occur in minute quantities. Because of their impurities, diamonds are classified into two types. Type I and Type II.
Type I - Nitrogen occur as the major impurity up to 0.2%.
Type II - Do not contain nitrogen as a significant impurity. Type II diamonds also contain nitrogen but does not affect the physical properties.
Type Ib transform into Type Ia.
The majority of natural diamonds are Type Ia.
Type Ia diamonds are diamonds that contain nitrogen atoms as pairs or small groups called aggregates.
Type IaA- diamonds contain aggregates of 2 nitrogen atoms
Type IaB - or groups of 3 or 4 nitrogens called aggregates with vacancies. Groups of 3 surrounding a vacancy are called N3 color centers.
Sunday, May 18, 2014
Electromagnetic Spectrum
The electromagnetic spectrum is the range of all possible frequencies of electromagnetic radiation.The "electromagnetic spectrum" of an object has a different meaning, and is instead the characteristic distribution of electromagnetic radiation emitted or absorbed by that particular object.
The electromagnetic spectrum extends from below the low frequencies used for modern radio communication to gamma radiation at the short-wavelength(high-frequency) end, thereby covering wavelengths from thousands of kilometers down to a fraction of the size of an atom. The limit for long wavelengths is the size of the universe itself, while it is thought that the short wavelength limit is in the vicinity of the Planck length,although in principle the spectrum is infinite and continuous.
Most parts of the electromagnetic spectrum are used in science for spectroscopic and other probing interactions, as ways to study and characterize matter. In addition, radiation from various parts of the spectrum has found many other uses for communications and manufacturing (see electromagnetic radiation for more applications).
Luminescence
Luminescence - describes emission of cold non incandescent light by a substance. A type of luminescence is fluorescence and phosphorescence.
Color Centers
Color Centers are defects in the crystal structure that absorbs light. These defects may occur during growth, or by irradiation. This irradiation may be natural or artificial.
Examples of color centers include:
Vibronic occur centers such as that in type IaA/B diamonds.
Examples of color centers include:
Vibronic occur centers such as that in type IaA/B diamonds.
Type II Diamonds
Type II diamonds have no significant amount of nitrogen
Type IIa - contain no significant impurities. Pure Type IIa diamonds have no sharp absorption in the visible region.
Type IIa diamonds which are capable of conducting electricity
Type IIb - boron atoms are present as an impurity
Type IIb diamonds which are not able to conduct electricity.
Type IIa - contain no significant impurities. Pure Type IIa diamonds have no sharp absorption in the visible region.
Type IIa diamonds which are capable of conducting electricity
Type IIb - boron atoms are present as an impurity
Type IIb diamonds which are not able to conduct electricity.
Type I Diamonds
Type Ia - Nitrogen atoms are present as pairs or small groups called aggregates
Type Ib - Nitrogen atoms are present as isolated atoms
Type IaA - Contain aggregates of two nitrogen atoms, A aggregates
Type IaB - Nitrogen is present as aggregates of four nitrogen toms surrounding a 'vacancy' B aggregates. (Most diamonds containing some nitrogen in the B form also have aggregates of three nitrogen atoms surrounding a vacancy.
It is common to find many diamonds with a mixture of A and B aggregates. These are described as type IaA/B or IaAB. Type IaAB - Contain both the A and B forms of nitrogen
Type Ib - Nitrogen atoms are present as isolated atoms
Type IaA - Contain aggregates of two nitrogen atoms, A aggregates
Type IaB - Nitrogen is present as aggregates of four nitrogen toms surrounding a 'vacancy' B aggregates. (Most diamonds containing some nitrogen in the B form also have aggregates of three nitrogen atoms surrounding a vacancy.
It is common to find many diamonds with a mixture of A and B aggregates. These are described as type IaA/B or IaAB. Type IaAB - Contain both the A and B forms of nitrogen
Tuesday, May 13, 2014
Birefringence
Birefringence is the optical property of a material having a refractive index that depends on the polarization and propagation direction of light.
Polarization is a property of waves that can oscillate with more than one direction.
Polarization is a property of waves that can oscillate with more than one direction.
Saturday, May 03, 2014
Volcanoes
A volcano is a rupture) on the crust) of a planetary mass object, such as the Earth, which allows hot lava, volcanic ash, and gases to escape from a magma chamber below the surface.
Earth's volcanoes occur because the planet's crust is broken into 17 major, rigid tectonic plates that float on a hotter, softer layer in the Earth's mantle. Therefore, on Earth, volcanoes are generally found where tectonic plates are diverging or converging. For example, a mid-oceanic ridge, such as the Mid-Atlantic Ridge, has volcanoes caused by divergent tectonic plates pulling apart; the Pacific Ring of Fire has volcanoes caused by convergent tectonic plates coming together. Volcanoes can also form where there is stretching and thinning of the crust's interior plates, e.g., in the East African Rift and the Wells Gray-Clearwater volcanic field and Rio Grande Rift in North America. This type of volcanism falls under the umbrella of "plate hypothesis" volcanism. Volcanism away from plate boundaries has also been explained as mantle plumes. These so-called "hotspots", for example Hawaii, are postulated to arise from upwelling diapirs with magma from the core–mantle boundary, 3,000 km deep in the Earth. Volcanoes are usually not created where two tectonic plates slide past one another.
Earth's volcanoes occur because the planet's crust is broken into 17 major, rigid tectonic plates that float on a hotter, softer layer in the Earth's mantle. Therefore, on Earth, volcanoes are generally found where tectonic plates are diverging or converging. For example, a mid-oceanic ridge, such as the Mid-Atlantic Ridge, has volcanoes caused by divergent tectonic plates pulling apart; the Pacific Ring of Fire has volcanoes caused by convergent tectonic plates coming together. Volcanoes can also form where there is stretching and thinning of the crust's interior plates, e.g., in the East African Rift and the Wells Gray-Clearwater volcanic field and Rio Grande Rift in North America. This type of volcanism falls under the umbrella of "plate hypothesis" volcanism. Volcanism away from plate boundaries has also been explained as mantle plumes. These so-called "hotspots", for example Hawaii, are postulated to arise from upwelling diapirs with magma from the core–mantle boundary, 3,000 km deep in the Earth. Volcanoes are usually not created where two tectonic plates slide past one another.
Extinct Volcanoes
An active volcano is a volcano that has had at least one eruption during the past 10,000 years. An active volcano might be erupting or dormant.
An erupting volcano is an active volcano that is having an eruption...
A dormant volcano is an active volcano that is not erupting, but supposed to erupt again.
An extinct volcano has not had an eruption for at least 10,000 years and is not expected to erupt again in a comparable time scale of the future.
Extinct Volcano
There are three ways to describe a volcano’s activity; there can be active, dormant, or extinct volcanoes. Active volcanoes have erupted recently. A dormant volcano isn’t erupting right now, but vulcanologists expect it could erupt at any time. Extinct volcanoes haven’t erupted for tens of thousands of years, and aren’t expected to erupt again.
Some volcanoes look extinct, but it might just be a long time since they’ve erupted. For example, the Yellowstone Caldera in Yellowstone National Park hasn’t had a violent eruption in about 640,000 years, but scientists think it’s still active. There has been minor activity and lava flows as recently as 10,000 years ago. The region also has regular minor earthquakes and ground is lifting up in some areas, so scientists think that’s it’s still an active volcano.
Thursday, May 01, 2014
Crystals and Color
How do crystals get their color? The presence of different chemicals causes the variety of colors to different gemstones. Many gems are simply quartz crystals colored by the environments to which they are exposed. Amethyst gets its color from iron found at specific points in the crystalline structure. Topaz is an aluminium silicate - it comes in many colors due to the presence of different chemicals. The color of any compound (whether or not it is a crystal) depends on how the atoms and or molecules absorb light. Normally white light (what comes out of light bulbs) is considered to have all wavelengths (colors) of light in it. If you pass a white light through a colored compound some of the light is absorbed (we don't see the color which is absorbed, but we see the rest of the light) as it is reflected off the surface. This gives rise to the idea of "Complementary Colors". If a compound absorbs light of a certain color the compound appears to be the complimentary color. Here is a table of colors and their compliments:
So if you have a crystal which absorbs red light, it will appear green and if the crystal absorbs green light, it will appear red.
So if you have a crystal which absorbs red light, it will appear green and if the crystal absorbs green light, it will appear red.
Molecule
Molecule - a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction.
Molecules form when two or more atoms form chemical bonds with each other. It doesn't matter if the atoms are the same or are different from each other.
Molecules Versus Compounds Molecules made up of two or more elements are called compounds. Water, calcium oxide, and glucose are molecules that are compounds. All compounds are molecules; not all molecules are compounds.
Molecules form when two or more atoms form chemical bonds with each other. It doesn't matter if the atoms are the same or are different from each other.
Molecules Versus Compounds Molecules made up of two or more elements are called compounds. Water, calcium oxide, and glucose are molecules that are compounds. All compounds are molecules; not all molecules are compounds.
What Is Not a Molecule?Single atoms of elements are not molecules. A single oxygen, O, is not a molecule. When oxygen bonds to itself (e.g., O2, O3) or to another element (e.g., carbon dioxide or CO2), molecules are formed.
What is a Mineral
Minerals can not be grown. Minerals are mined. Minerals are an energy source. Any naturally occurring inorganic substance having a definite chemical composition and characteristic crystalline structure, color, and hardness. There are approximately 4000 varieties of minerals which can be classified into one of eight major mineral classes similar to how biologists classify living organisms into groups such as plants, animals, fungi, and bacteria.
Mineral Classification
Minerals, however, are classified into groups by their composition and crystal structure. Ninety five percent of the rocks found in Earth’s crust contain minerals known as silicates. Silicates are the most abundant minerals and make up about 25% of all known minerals. The structure of all silicates is based on a silicon atom surrounded by four oxygen atoms. Silicates can be further classified into six subclasses differentiated by their structure. Common silicates include quartz, feldspars, pyroxene and olivine.
Mineral Classification
Minerals, however, are classified into groups by their composition and crystal structure. Ninety five percent of the rocks found in Earth’s crust contain minerals known as silicates. Silicates are the most abundant minerals and make up about 25% of all known minerals. The structure of all silicates is based on a silicon atom surrounded by four oxygen atoms. Silicates can be further classified into six subclasses differentiated by their structure. Common silicates include quartz, feldspars, pyroxene and olivine.
What is an Atom
An atom is the smallest unit of a chemical element. It is a fundamental piece of matter. A matter is anything that can be touched physically. Everything in the universe is made of matter except energy. So everything in the universe is made up of atoms.
An atom is made out of particles called subatomic particles: protons, neutrons, and electrons.
The electrons which are negatively charged fly around or orbit the nucleus which in the center of the atom and consists of the protons and neutrons which are negatively charged. Typically, the number of electrons and protons are equal.
Carbon element has 6 elections, 6 protons and 6 neutrons.
The negative electrons are attracted to the positive nucleus by the same electrical force which causes magnets to work. That's what keeps the atom together.





An atom is made out of particles called subatomic particles: protons, neutrons, and electrons.
The electrons which are negatively charged fly around or orbit the nucleus which in the center of the atom and consists of the protons and neutrons which are negatively charged. Typically, the number of electrons and protons are equal.
Carbon element has 6 elections, 6 protons and 6 neutrons.
The negative electrons are attracted to the positive nucleus by the same electrical force which causes magnets to work. That's what keeps the atom together.
What is a crystal and how is it formed?
A crystal is a solid geometric structure of a repeating pattern of molecules connecting it together. In crystals, a unit cell is repeated in exactly the same arrangement over and over throughout the entire material.
Crystals grow by a process called "nucleation". Nucleation can start with assisted nucleation or unassisted nucleation. It will continue to grow until an equilibrium is reached.
Crystals grow by a process called "nucleation". Nucleation can start with assisted nucleation or unassisted nucleation. It will continue to grow until an equilibrium is reached.
Why crystals grow at different rates in different directions is a very complicated question. If there is a highly attractive interaction (energetically speaking) along a certain direction of a crystal, then that direction will probably grow fast. However, it could also grow slowly, if that direction interacted strongly with the solvent; having strongly absorbed solvent on the surface of the crystal could block growth.


Refraction and Refractive Index
When light enters a liquid or solid substance from air its speed is greatly reduced. This bending effect is called refraction. Different substances slow the light to different speeds.
The more the light is slowed when it enters the material, the more the material will bend the light (the greater amount of refraction). Different materials tend to give different results for this refraction.
We can think of this as the optical density: the greater the slowing effect, the greater the optical density of the material. The greater the optical density, the greater the refractive power. It's too difficult to measure the speed of light through air, instead we measure it's bending effect.
Refraction is the change in direction in which light travels when it passes from one medium into another of differing optical density.
The Refractive Index of a diamond is 2.42 compared to quartz which is 1.54-1.56. Refraction is an important optical effect cause it has a diagnostic value. Refraction has a direct relationship to other optical properties such as reflection and brilliance.
Refractive Index is a number that describes how light propagates through that medium. The refractive index determines how much light is bent, or refracted, when entering a material.
The more the light is slowed when it enters the material, the more the material will bend the light (the greater amount of refraction). Different materials tend to give different results for this refraction.
We can think of this as the optical density: the greater the slowing effect, the greater the optical density of the material. The greater the optical density, the greater the refractive power. It's too difficult to measure the speed of light through air, instead we measure it's bending effect.
Refraction is the change in direction in which light travels when it passes from one medium into another of differing optical density.
The Refractive Index of a diamond is 2.42 compared to quartz which is 1.54-1.56. Refraction is an important optical effect cause it has a diagnostic value. Refraction has a direct relationship to other optical properties such as reflection and brilliance.
Refractive Index is a number that describes how light propagates through that medium. The refractive index determines how much light is bent, or refracted, when entering a material.
Wednesday, April 30, 2014
Cubic Crystal System
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
In geometry, a cube is a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex.
In geometry, a cube is a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex.
Diamond Cubic Structure
The diamond cubic crystal structure is a repeating pattern of 8 atoms that certain materials may adopt as they solidify. While the first known example was diamond, other elements in group 14 also adopt this structure, including α-tin, the semiconductors silicon and germanium, and silicon/germanium alloys in any proportion.
Diamond cubic is in the Fd3m space group, which follows the face-centered cubic bravais lattice. The lattice describes the repeat pattern; for diamond cubic crystals this lattice is "decorated" with a motif of two tetrahedrally bonded atoms in each primitive cell, separated by 1/4 of the width of the unit cell in each dimension.
Diamond cubic is in the Fd3m space group, which follows the face-centered cubic bravais lattice. The lattice describes the repeat pattern; for diamond cubic crystals this lattice is "decorated" with a motif of two tetrahedrally bonded atoms in each primitive cell, separated by 1/4 of the width of the unit cell in each dimension.
Crystal Twinning
What is crystal twining?
A process in which two or more crystals, or parts of crystals, assume orientations such that one may be brought to coincidence with the other byreflection across a plane or by rotation about an axis. Crystal twins represent a particularly symmetric kind of grain boundary; however, the Energy of the twin boundary is much lower than that of the general grain boundary because some of the atoms in the twin interface are in the correct positions relative to each other.
Crystal twinning is inter grown crystals that are formed in a symmetrical fashion.
It is caused by stress during the growth or temperature/pressure. These inter grown crystals are called twinned crystals.
Twinning is important to recognize cause it's helps identification of minerals in nature.
A crystal structure may contain a change in repeat-direction, to form a twin. Two or more parts of a twinned crystal structure are not related by the normal symmetry of that structure. They are related by a twin reflection plane or axis, or both. The twinning of a structure may be an accident of growth, or caused by stress or change in temperature during growth.
Crystal twinning occurs when two separate crystals share some of the same crystal lattice points in a symmetrical manner. The result is an inter-growth of two separate crystals in a variety of specific configurations. A twin boundary or composition surface separates the two crystals. Crystallographers classify twinned crystals by a number of twin laws. These twin laws are specific to the crystal system. The type of twinning can be a diagnostic tool in mineral identification.
A process in which two or more crystals, or parts of crystals, assume orientations such that one may be brought to coincidence with the other byreflection across a plane or by rotation about an axis. Crystal twins represent a particularly symmetric kind of grain boundary; however, the Energy of the twin boundary is much lower than that of the general grain boundary because some of the atoms in the twin interface are in the correct positions relative to each other.
The formation in a single crystal of regions of regularly changed orientation of the crystal structure. The structures of twin formations are eithermirror images of the atomic structure of the parent crystal (matrix) in a certain plane (the twinning plane) or are formed by rotation of the matrixstructure about the crystallographic axis (twinning axis) to an angle that is constant for a given material or by other symmetry transformations. The pair made up of the matrix and the twin formation is called the twin.
Twinning takes place during crystal growth because of violations in the packing of atoms during the growth of the atom layer on the nucleus or onthe formed crystal (stacking faults) and the intergrowth of neighboring nuclei (growth twins; see Figure l). It also takes place because ofdeformation upon mechanical action on the crystal, such as the impact of an indentor, tension, compression, twisting, bending (mechanical twins);rapid thermal expansion and contraction; heating of deformed crystals (recrystallization twins); and transition from one crystal modification toanother.
Crystal twinning is inter grown crystals that are formed in a symmetrical fashion.
It is caused by stress during the growth or temperature/pressure. These inter grown crystals are called twinned crystals.
Twinning is important to recognize cause it's helps identification of minerals in nature.
A crystal structure may contain a change in repeat-direction, to form a twin. Two or more parts of a twinned crystal structure are not related by the normal symmetry of that structure. They are related by a twin reflection plane or axis, or both. The twinning of a structure may be an accident of growth, or caused by stress or change in temperature during growth.
Crystal twinning occurs when two separate crystals share some of the same crystal lattice points in a symmetrical manner. The result is an inter-growth of two separate crystals in a variety of specific configurations. A twin boundary or composition surface separates the two crystals. Crystallographers classify twinned crystals by a number of twin laws. These twin laws are specific to the crystal system. The type of twinning can be a diagnostic tool in mineral identification.
Chemical Element
A chemical element is a pure chemical substance consisting of a single type of atom distinguished by its atomic number, which is the number of protons in its atomic nucleus.
Polymorphism and Allotropy
It's the ability of a chemical substance to exist in more than one alternative crystal structure. It is related to allotropy and is described by crystal habit, amorphous, or crystal defect.
Polymorphism and Allotropy are same thing. Polymorphism is used for compounds and the allotropy is reserved for elements.
Allotropy or allotropism (from Greek ἄλλος (allos), meaning "other", and τρόπος (tropos), meaning "manner, form") is the property of some chemical elements to exist in two or more different forms, known as allotropes of these elements. Allotropes are different structural modifications of an element; the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (where the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (where the carbon atoms are bonded together in sheets of a hexagonal lattice).



Polymorphism and Allotropy are same thing. Polymorphism is used for compounds and the allotropy is reserved for elements.
Allotropy or allotropism (from Greek ἄλλος (allos), meaning "other", and τρόπος (tropos), meaning "manner, form") is the property of some chemical elements to exist in two or more different forms, known as allotropes of these elements. Allotropes are different structural modifications of an element; the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (where the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (where the carbon atoms are bonded together in sheets of a hexagonal lattice).



Sunday, April 27, 2014
Lattice System
Lattice System
These lattice systems are a grouping of crystal structures according to the axial system used to describe their lattice. Each lattice system consists of a set of three axes in a particular geometric arrangement. There are seven lattice systems. They are similar to but not quite the same as the seven crystal systems and the six crystal families.
These lattice systems are a grouping of crystal structures according to the axial system used to describe their lattice. Each lattice system consists of a set of three axes in a particular geometric arrangement. There are seven lattice systems. They are similar to but not quite the same as the seven crystal systems and the six crystal families.
The simplest and most symmetric, the cubic (or isometric) system, has the symmetry of a cube, that is, it exhibits four threefold rotational axes oriented at 109.5° (the tetrahedral angle) with respect to each other. These threefold axes lie along the body diagonals of the cube. The other six lattice systems, are hexagonal, tetragonal, rhombohedral (often confused with the trigonal crystal system),orthorhombic, monoclinic and triclinic.
| The 7 lattice systems (From least to most symmetric) | The 14 Bravais Lattices | |||
| 1. triclinic (none) |  | |||
| 2. monoclinic (1 diad) | simple | base-centered | ||
 |  | |||
| 3. orthorhombic (3 perpendicular diads) | simple | base-centered | body-centered | face-centered |
 |  |  |  | |
| 4. rhombohedral (1 triad) |  | |||
| 5. tetragonal (1 tetrad) | simple | body-centered | ||
 |  | |||
| 6. hexagonal (1 hexad) | 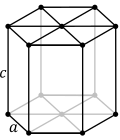 | |||
| 7. cubic (4 triads) | simple (SC) | body-centered (bcc) | face-centered (fcc) | |
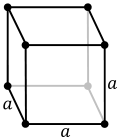 | 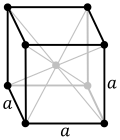 |  | ||
Subscribe to:
Posts (Atom)
