In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
In geometry, a cube is a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex.
Wednesday, April 30, 2014
Diamond Cubic Structure
The diamond cubic crystal structure is a repeating pattern of 8 atoms that certain materials may adopt as they solidify. While the first known example was diamond, other elements in group 14 also adopt this structure, including α-tin, the semiconductors silicon and germanium, and silicon/germanium alloys in any proportion.
Diamond cubic is in the Fd3m space group, which follows the face-centered cubic bravais lattice. The lattice describes the repeat pattern; for diamond cubic crystals this lattice is "decorated" with a motif of two tetrahedrally bonded atoms in each primitive cell, separated by 1/4 of the width of the unit cell in each dimension.
Diamond cubic is in the Fd3m space group, which follows the face-centered cubic bravais lattice. The lattice describes the repeat pattern; for diamond cubic crystals this lattice is "decorated" with a motif of two tetrahedrally bonded atoms in each primitive cell, separated by 1/4 of the width of the unit cell in each dimension.
Crystal Twinning
What is crystal twining?
A process in which two or more crystals, or parts of crystals, assume orientations such that one may be brought to coincidence with the other byreflection across a plane or by rotation about an axis. Crystal twins represent a particularly symmetric kind of grain boundary; however, the Energy of the twin boundary is much lower than that of the general grain boundary because some of the atoms in the twin interface are in the correct positions relative to each other.
Crystal twinning is inter grown crystals that are formed in a symmetrical fashion.
It is caused by stress during the growth or temperature/pressure. These inter grown crystals are called twinned crystals.
Twinning is important to recognize cause it's helps identification of minerals in nature.
A crystal structure may contain a change in repeat-direction, to form a twin. Two or more parts of a twinned crystal structure are not related by the normal symmetry of that structure. They are related by a twin reflection plane or axis, or both. The twinning of a structure may be an accident of growth, or caused by stress or change in temperature during growth.
Crystal twinning occurs when two separate crystals share some of the same crystal lattice points in a symmetrical manner. The result is an inter-growth of two separate crystals in a variety of specific configurations. A twin boundary or composition surface separates the two crystals. Crystallographers classify twinned crystals by a number of twin laws. These twin laws are specific to the crystal system. The type of twinning can be a diagnostic tool in mineral identification.
A process in which two or more crystals, or parts of crystals, assume orientations such that one may be brought to coincidence with the other byreflection across a plane or by rotation about an axis. Crystal twins represent a particularly symmetric kind of grain boundary; however, the Energy of the twin boundary is much lower than that of the general grain boundary because some of the atoms in the twin interface are in the correct positions relative to each other.
The formation in a single crystal of regions of regularly changed orientation of the crystal structure. The structures of twin formations are eithermirror images of the atomic structure of the parent crystal (matrix) in a certain plane (the twinning plane) or are formed by rotation of the matrixstructure about the crystallographic axis (twinning axis) to an angle that is constant for a given material or by other symmetry transformations. The pair made up of the matrix and the twin formation is called the twin.
Twinning takes place during crystal growth because of violations in the packing of atoms during the growth of the atom layer on the nucleus or onthe formed crystal (stacking faults) and the intergrowth of neighboring nuclei (growth twins; see Figure l). It also takes place because ofdeformation upon mechanical action on the crystal, such as the impact of an indentor, tension, compression, twisting, bending (mechanical twins);rapid thermal expansion and contraction; heating of deformed crystals (recrystallization twins); and transition from one crystal modification toanother.
Crystal twinning is inter grown crystals that are formed in a symmetrical fashion.
It is caused by stress during the growth or temperature/pressure. These inter grown crystals are called twinned crystals.
Twinning is important to recognize cause it's helps identification of minerals in nature.
A crystal structure may contain a change in repeat-direction, to form a twin. Two or more parts of a twinned crystal structure are not related by the normal symmetry of that structure. They are related by a twin reflection plane or axis, or both. The twinning of a structure may be an accident of growth, or caused by stress or change in temperature during growth.
Crystal twinning occurs when two separate crystals share some of the same crystal lattice points in a symmetrical manner. The result is an inter-growth of two separate crystals in a variety of specific configurations. A twin boundary or composition surface separates the two crystals. Crystallographers classify twinned crystals by a number of twin laws. These twin laws are specific to the crystal system. The type of twinning can be a diagnostic tool in mineral identification.
Chemical Element
A chemical element is a pure chemical substance consisting of a single type of atom distinguished by its atomic number, which is the number of protons in its atomic nucleus.
Polymorphism and Allotropy
It's the ability of a chemical substance to exist in more than one alternative crystal structure. It is related to allotropy and is described by crystal habit, amorphous, or crystal defect.
Polymorphism and Allotropy are same thing. Polymorphism is used for compounds and the allotropy is reserved for elements.
Allotropy or allotropism (from Greek ἄλλος (allos), meaning "other", and τρόπος (tropos), meaning "manner, form") is the property of some chemical elements to exist in two or more different forms, known as allotropes of these elements. Allotropes are different structural modifications of an element; the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (where the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (where the carbon atoms are bonded together in sheets of a hexagonal lattice).



Polymorphism and Allotropy are same thing. Polymorphism is used for compounds and the allotropy is reserved for elements.
Allotropy or allotropism (from Greek ἄλλος (allos), meaning "other", and τρόπος (tropos), meaning "manner, form") is the property of some chemical elements to exist in two or more different forms, known as allotropes of these elements. Allotropes are different structural modifications of an element; the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (where the carbon atoms are bonded together in a tetrahedral lattice arrangement), graphite (where the carbon atoms are bonded together in sheets of a hexagonal lattice).



Sunday, April 27, 2014
Lattice System
Lattice System
These lattice systems are a grouping of crystal structures according to the axial system used to describe their lattice. Each lattice system consists of a set of three axes in a particular geometric arrangement. There are seven lattice systems. They are similar to but not quite the same as the seven crystal systems and the six crystal families.
These lattice systems are a grouping of crystal structures according to the axial system used to describe their lattice. Each lattice system consists of a set of three axes in a particular geometric arrangement. There are seven lattice systems. They are similar to but not quite the same as the seven crystal systems and the six crystal families.
The simplest and most symmetric, the cubic (or isometric) system, has the symmetry of a cube, that is, it exhibits four threefold rotational axes oriented at 109.5° (the tetrahedral angle) with respect to each other. These threefold axes lie along the body diagonals of the cube. The other six lattice systems, are hexagonal, tetragonal, rhombohedral (often confused with the trigonal crystal system),orthorhombic, monoclinic and triclinic.
| The 7 lattice systems (From least to most symmetric) | The 14 Bravais Lattices | |||
| 1. triclinic (none) |  | |||
| 2. monoclinic (1 diad) | simple | base-centered | ||
 |  | |||
| 3. orthorhombic (3 perpendicular diads) | simple | base-centered | body-centered | face-centered |
 |  |  |  | |
| 4. rhombohedral (1 triad) |  | |||
| 5. tetragonal (1 tetrad) | simple | body-centered | ||
 |  | |||
| 6. hexagonal (1 hexad) | 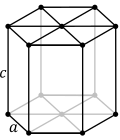 | |||
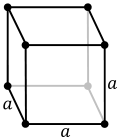
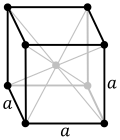
| 7. cubic (4 triads) | simple (SC) | body-centered (bcc) | face-centered (fcc) | |
 |  |  | ||
Face Centered Cubic
Unit Cell
The crystal structure of a material (the arrangement of atoms within a given type of crystal) can be described in terms of its unit cell. The unit cell is a small box containing one or more atoms arranged in 3-dimensions. The unit cells stacked in three-dimensional space describe the bulk arrangement of atoms of the crystal. The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges (a,b and c) and the angles between them (alpha, beta and gamma), while the positions of the atoms inside the unit cell are described by the set of atomic positions (xi , yi , zi) measured from a lattice point. Commonly, atomic positions are represented in terms of fractional coordinates, relative to the unit cell lengths.
The atom positions within the unit cell can be calculated through application of symmetry operations to the asymmetric unit. The asymmetric unit refers to the smallest possible occupation of space within the unit cell. This does not, however imply that the entirety of the asymmetric unit must lie within the boundaries of the unit cell. Symmetric transformations of atom positions are calculated from the space group of the crystal structure, and this is usually a black box operation performed by computer programs. However, manual calculation of the atomic positions within the unit cell can be performed from the asymmetric unit, through the application of the symmetry operators described within the 'International Tables for Crystallography: Volume A.



Simple Cubic Body Centered Cubic Face Centered Cubic
The crystal structure of a material (the arrangement of atoms within a given type of crystal) can be described in terms of its unit cell. The unit cell is a small box containing one or more atoms arranged in 3-dimensions. The unit cells stacked in three-dimensional space describe the bulk arrangement of atoms of the crystal. The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges (a,b and c) and the angles between them (alpha, beta and gamma), while the positions of the atoms inside the unit cell are described by the set of atomic positions (xi , yi , zi) measured from a lattice point. Commonly, atomic positions are represented in terms of fractional coordinates, relative to the unit cell lengths.
The atom positions within the unit cell can be calculated through application of symmetry operations to the asymmetric unit. The asymmetric unit refers to the smallest possible occupation of space within the unit cell. This does not, however imply that the entirety of the asymmetric unit must lie within the boundaries of the unit cell. Symmetric transformations of atom positions are calculated from the space group of the crystal structure, and this is usually a black box operation performed by computer programs. However, manual calculation of the atomic positions within the unit cell can be performed from the asymmetric unit, through the application of the symmetry operators described within the 'International Tables for Crystallography: Volume A.
Simple Cubic Body Centered Cubic Face Centered Cubic
Polycrystalline
Polycrystalline materials are solids that are composed of many crystallites of varying size and orientation. The variation in direction can be random (called random texture) or directed, possibly due to growth and processing conditions

Friday, April 25, 2014
Crystallography
Crystallography is the science that examines the arrangement of atoms in solids (see crystal structure). This involves measuring the angles of crystal faces relative to theoretical reference axes (crystallographic axes), and establishing the symmetry of the crystal in question.
Crystallographic Axis
One of the imaginary reference lines passing through the center of anideal crystal, designated a, b, or c.
Crystal Lattice
Crystal lattice is the periodic and systematic arrangement of atoms that are found in crystals with the exception of amorphous solids and gases. In the simplest of terms, the crystal lattice can be considered as the points of intersection between straight lines in a three-dimensional network. The physical properties of crystals like cleavage, electronic band structure and optical transparency are predominantly governed by the crystal lattice. A unit cell is the smallest component of the crystal lattice and describes the arrangement of atoms in a crystal. The unit cell is characterized by its lattice parameters which consist of the length of the cell edges and the angles between them.

There are seven crystal lattice systems namely monoclinic, triclinic, orthorhombic, rhombohedral, tetragonal, hexagonal and cubic. All these seven crystal lattice have their own variants giving rise to a total of 14 Bravais Lattices. Among these seven lattice systems, the cubic system exhibits the symmetry of a cube is considered to be the simplest and also the most symmetric of all crystal lattices. There are three different types of cubical crystal lattice systems namely Simple Cubic (SC), Body-Centered Cubic (CBC) and the Face Centered Cubic (FCC). As the name indicates, Simple Cubic is the simplest form of the structure. The simple cubic unit cell has one lattice point at each of the eight corners of the cube. The number of lattice points is usually denoted by Z and for a SC, the value of Z is 1. The packing efficiency of a lattice is defined as the ratio of total volume of the cell occupied by the host atom to the volume of the cell. Usually, crystals with higher packing efficiency are preferred because of their favorable properties. This packing efficiency for SC works has been reported to be around 52%.
Cubic System
In cubic system there are three reference axes all of the same length and all at right angles to each other.
Wednesday, April 23, 2014
U.S. Diamond Mines
One Active Mine Today ...
The United States only has two locations that have been operated as commercial diamond mines. One was the Kelsey Lake Diamond Mine near Fort Collins, Colorado. It produced small amounts of diamonds between 1996 and 2002 when the mine was closed due to legal problems.
The second is at Crater of Diamonds State Park near Murfreesboro, Arkansas. It was worked as a commercial diamond mine in the early 1900s but closed because the deposit was subeconomic. It has since been operated by the State of Arkansas as a tourist "pay-to-dig mine" where anyone can pay a fee, look for diamonds and keep any that they find. A few hundred carats of diamonds are found there each year.
Diamonds, diamond indicator mineral assemblages and potentially diamondiferous rocks have been found at hundreds of locations within the United States. One or more of these locations could become commercial and make the United States a significant producer of gem-quality diamonds.
Crater of Diamonds - The Only Active U. S. Mine
Although millions of carats of diamonds are consumed each year in the United States, only a few hundred carats are domestically produced. The only active diamond mine in the country is the Crater of Diamonds Mine near Murfreesboro, Pike County, Arkansas. There, recreational prospectors have been finding a few hundred carats of diamonds per year since the early 1970s. Most of the stones are white, yellow and brown in color but a wide range of diamond colors has been found at the mine. See photo at top right.
Diamonds in Volcanoes
If diamonds are formed deep down the earth, how do diamonds get to the surface?
Volcanoes - that's how. When volcanoes erupt, sometimes diamonds are thrown out of the top in the lava or are left in pipe, the central, vertical part of the volcano. The best place to find diamonds is in the center of an extinct volcano, in a type of rock called kimberlite.
Diamonds are brought to the surface from the mantle in a rare type of magma called kimberlite and erupted at a rare type of volcanic vent called a diatreme or pipe. Kimberlite is a gas-rich, potassicultramafic igneous rock that contains the minerals olivine, phlogopite, diopside, serpentine, calcite, and minor amounts of apatite, magnetite, chromite, garnet, diamond, and other upper mantle minerals. Upper mantle xenoliths are found in some kimberlite and provide clues to the magma's origin. The source depth for kimberlite magmas is estimated at 200 km, more than twice as deep as the source region for most magmas. At a depth of 200 km the pressure is 60,000 times greater than the surface and the temperature is about 1500 C. Kimberlite magmas are rich in carbon dioxide and water which brings the magma quickly and violently to the surface. Most kimberlites occur as multiple intrusive events. Kimberlite was named for the rock associated with diamonds in Kimberley, South Africa.
Kimberlite magmas form "pipes" as they erupt. A tuff cone is at the surface and formed by base-surge deposits. In the subsurface, a funnel-shaped body narrows to a depth of hundreds of meters. The pipe (also called a diatreme) is filled with kimberlite, with or without diamonds (only 1 in 5 of the pipes at Kimberley contain diamonds).
Just how many diamonds are needed to make aa pipe economical? Some South African mines operate at 25 carats of diamond per 100 cubic meters of rock or about 2 grams of diamonds per 100 tons of rock. Because diamond has a specific gravity of 3.5 grams per cubic centimeter, 1 cubic centimeter of diamond weighs 16 carats. Picture a giant 100-ton ore truck full of kimberlite - that truck contains only half of a cubic centimeter of diamonds! Only about 35% of those diamonds are gem quality.
Kimberlite magmas form "pipes" as they erupt. A tuff cone is at the surface and formed by base-surge deposits. In the subsurface, a funnel-shaped body narrows to a depth of hundreds of meters. The pipe (also called a diatreme) is filled with kimberlite, with or without diamonds (only 1 in 5 of the pipes at Kimberley contain diamonds).
Just how many diamonds are needed to make aa pipe economical? Some South African mines operate at 25 carats of diamond per 100 cubic meters of rock or about 2 grams of diamonds per 100 tons of rock. Because diamond has a specific gravity of 3.5 grams per cubic centimeter, 1 cubic centimeter of diamond weighs 16 carats. Picture a giant 100-ton ore truck full of kimberlite - that truck contains only half of a cubic centimeter of diamonds! Only about 35% of those diamonds are gem quality.
Fluorescence
What is a Fluorescent Mineral?
The wavelength of light released from a fluorescent mineral is often distinctly different from the wavelength of the incident light. This produces a visible change in the color of the mineral. This "glow" continues as long as the mineral is illuminated with light of the proper wavelength.
All minerals have the ability to reflect light. That is what makes them visible to the human eye. A few minerals have an interesting physical property known as "fluorescence". These minerals have the ability to temporarily absorb a small amount of light and an instant later release a small amount of light of a different wavelength. This change in wavelength causes a temporary color change of the mineral in the eye of a human observer.
The color change of fluorescent minerals is most spectacular when they are illuminated in darkness by ultraviolet light (which is not visible to humans) and they release visible light. The photograph above is an example of this phenomenon.
Fluorite: The Original "Fluorescent Mineral"
One of the first people to observe fluorescence in minerals was George Gabriel Stokes in 1852. He noted the ability of fluorite to produce a blue glow when illuminated with invisible light "beyond the violet end of the spectrum". He called this phenomenon "fluorescence" after the mineral fluorite. The name has gained wide acceptance in mineralogy, gemology, biology, optics, commercial lighting and many other fields.
Many specimens of fluorite have a strong enough fluorescence that the observer can take them outside, hold them in sunlight then move them into shade and see a color change. Only a few minerals have this level of fluorescence. Fluorite typically glows a blue-violet color under short-wave and long-wave light. Some specimens are known to glow a cream or white color. Many specimens do not fluoresce. Fluorescence in fluorite is thought to be caused by the presence of yttrium, europium, samarium or organic material as activators.
The color change of fluorescent minerals is most spectacular when they are illuminated in darkness by ultraviolet light (which is not visible to humans) and they release visible light. The photograph above is an example of this phenomenon.
Fluorite: The Original "Fluorescent Mineral"
One of the first people to observe fluorescence in minerals was George Gabriel Stokes in 1852. He noted the ability of fluorite to produce a blue glow when illuminated with invisible light "beyond the violet end of the spectrum". He called this phenomenon "fluorescence" after the mineral fluorite. The name has gained wide acceptance in mineralogy, gemology, biology, optics, commercial lighting and many other fields.
Many specimens of fluorite have a strong enough fluorescence that the observer can take them outside, hold them in sunlight then move them into shade and see a color change. Only a few minerals have this level of fluorescence. Fluorite typically glows a blue-violet color under short-wave and long-wave light. Some specimens are known to glow a cream or white color. Many specimens do not fluoresce. Fluorescence in fluorite is thought to be caused by the presence of yttrium, europium, samarium or organic material as activators.
Fluorescence in More Detail
Fluorescence in minerals occurs when a specimen is illuminated with specific wavelengths of light. Ultraviolet light, x-rays and cathode rays are the typical types of light that trigger fluorescence. These types of light have the ability to excite susceptible electrons within the atomic structure of the mineral. These excited electrons temporarily jump up to a higher orbital within the mineral's atomic structure. When those electrons fall back down to their original orbital a small amount of energy is released in the form of light. This release of light is known as fluorescence.
Fluorescence in minerals occurs when a specimen is illuminated with specific wavelengths of light. Ultraviolet light, x-rays and cathode rays are the typical types of light that trigger fluorescence. These types of light have the ability to excite susceptible electrons within the atomic structure of the mineral. These excited electrons temporarily jump up to a higher orbital within the mineral's atomic structure. When those electrons fall back down to their original orbital a small amount of energy is released in the form of light. This release of light is known as fluorescence.
The wavelength of light released from a fluorescent mineral is often distinctly different from the wavelength of the incident light. This produces a visible change in the color of the mineral. This "glow" continues as long as the mineral is illuminated with light of the proper wavelength.
How Many Minerals Fluoresce in UV Light?
Most minerals do not fluoresce. Only about 15% of minerals have this ability and every specimen of those minerals does not fluoresce. [2] Fluorescence usually occurs when specific impurities known as "activators" are present within the mineral. These activators are typically cations of metals such as: tungsten, molybdenum, lead, boron, titanium, manganese, uranium and chromium. Rare earth elements such as europium, terbium, dysprosium, and yttrium are also known to contribute to the fluorescence phenomenon. Fluorescence can also be caused by crystal structural defects or organic impurities.
In addition to "activator" impurities, some impurities have a dampening effect on fluorescence. If iron or copper are present as impurities they can reduce or eliminate fluorescence. Furthermore, if the activator mineral is present in large amounts, that can reduce the fluorescence effect.
Most minerals fluoresce a single color. Other minerals have multiple colors of fluorescence. Calcite has been known to fluoresce red, blue, white, pink, green and orange. Some minerals are known to exhibit multiple colors of fluorescence in a single specimen. These can be banded minerals that exhibit several stages of growth from parent solutions with changing compositions. Many minerals fluoresce one color under short-wave UV light and another color under long-wave UV light.
Lamps for Viewing Fluorescent Minerals
The lamps used to locate and study fluorescent minerals are very different from the ultraviolet lamps (called "black lights") sold in novelty stores. The novelty store lamps are not suitable for mineral studies for two reasons: 1) they emit long-wave ultraviolet light (most fluorescent minerals respond to short-wave ultraviolet); and, 2) they emit a significant amount of visible light which interferes with accurate observation, but is not a problem for novelty use.
The scientific-grade lamps used for mineral studies have a filter that blocks most of the visible light that will interfere with observation. These filters are very expensive and are partly responsible for the significantly higher price of scientific lamps.
Most minerals do not fluoresce. Only about 15% of minerals have this ability and every specimen of those minerals does not fluoresce. [2] Fluorescence usually occurs when specific impurities known as "activators" are present within the mineral. These activators are typically cations of metals such as: tungsten, molybdenum, lead, boron, titanium, manganese, uranium and chromium. Rare earth elements such as europium, terbium, dysprosium, and yttrium are also known to contribute to the fluorescence phenomenon. Fluorescence can also be caused by crystal structural defects or organic impurities.
In addition to "activator" impurities, some impurities have a dampening effect on fluorescence. If iron or copper are present as impurities they can reduce or eliminate fluorescence. Furthermore, if the activator mineral is present in large amounts, that can reduce the fluorescence effect.
Most minerals fluoresce a single color. Other minerals have multiple colors of fluorescence. Calcite has been known to fluoresce red, blue, white, pink, green and orange. Some minerals are known to exhibit multiple colors of fluorescence in a single specimen. These can be banded minerals that exhibit several stages of growth from parent solutions with changing compositions. Many minerals fluoresce one color under short-wave UV light and another color under long-wave UV light.
Lamps for Viewing Fluorescent Minerals
The lamps used to locate and study fluorescent minerals are very different from the ultraviolet lamps (called "black lights") sold in novelty stores. The novelty store lamps are not suitable for mineral studies for two reasons: 1) they emit long-wave ultraviolet light (most fluorescent minerals respond to short-wave ultraviolet); and, 2) they emit a significant amount of visible light which interferes with accurate observation, but is not a problem for novelty use.
The scientific-grade lamps used for mineral studies have a filter that blocks most of the visible light that will interfere with observation. These filters are very expensive and are partly responsible for the significantly higher price of scientific lamps.
Electrical Conductivity
Conductivity: diamond cannot conduct electricity as there are no electrons free to move between the layers and whereas graphite can conduct electricity because in graphite, each carbon atom is covalently bonded to 3 others and this leaves delocalized electron free to move and thus graphite can conduct.
Host Rock
What is a host rock?
The host rock is normally defined as most dominant host rock where mineralization occurs. A body of rock serving as a host for other rock or for mineral deposits. Any rock which ore deposits occur.
Rock which serves as a host for other rocks or for mineral deposits.
The host rock is normally defined as most dominant host rock where mineralization occurs. A body of rock serving as a host for other rock or for mineral deposits. Any rock which ore deposits occur.
Rock which serves as a host for other rocks or for mineral deposits.
Sunday, April 20, 2014
Atomic Structure of a Diamond
Atomic structure is the way in which atoms are arranged in a molecule with specific details about protons,electron, and neutrons.
Graphite has a 2D arrangement in a trigonal planar shape, forming layers with weak intermolecular forces and the weak forces are easily broken. Diamond is hard because the covalent bonds between the carbon atoms are very strong and hold the atom firmly in its 3D shape.
Graphite has a 2D arrangement in a trigonal planar shape, forming layers with weak intermolecular forces and the weak forces are easily broken. Diamond is hard because the covalent bonds between the carbon atoms are very strong and hold the atom firmly in its 3D shape.
Diamond Hardness
Hardness is the ability of a material to resist abrasion when a pointed fragment of another substance is drawn across it without sufficient pressure to develop fracture or cleavage.
Differential structure is related to both the crystal and atomic structure of the diamond crystal.
Diamonds have differential hardness which means the hardness differs with the direction of scratching in certain gem materials. This differential hardness is due to the arrangement of the atomic bonding in crystal structures.
In various directions along the octahedron faces of diamond, hardness is great.
Although a diamond is the hardest know substance, the fact that it can be cut and polished is due to the phenomenon of differential hardness.
Damage by continual scratching is called abrasion. In the mineralogy world, only a diamond can scratch another diamond.
Differential structure is related to both the crystal and atomic structure of the diamond crystal.
Diamonds have differential hardness which means the hardness differs with the direction of scratching in certain gem materials. This differential hardness is due to the arrangement of the atomic bonding in crystal structures.
In various directions along the octahedron faces of diamond, hardness is great.
Although a diamond is the hardest know substance, the fact that it can be cut and polished is due to the phenomenon of differential hardness.
Damage by continual scratching is called abrasion. In the mineralogy world, only a diamond can scratch another diamond.
Thursday, April 17, 2014
River Deposit Mining
River Deposit Mining
Diamonds are a brittle substance despite it's hardness. It can shatter if struck by a sharp blow. In nature, diamond minerals need to be tough enough to survive in and gravel deposits. The minerals that are not tough enough to survive the continual shocks simply fracture, corrode away. Diamonds survive extremely well in this natural process.
Dredging - Large scale suction dredger and all material is examined for diamonds. Waste is discarded from the dredger into an area which already has been cleared.
Dredging is an excavation activity or operation usually carried out at least partly underwater, in shallow seas or fresh water areas with the purpose of gathering up bottom sediments and disposing of them at a different location. This technique is often used to keep waterways navigable.
Diverting Rivers - It is quite common in Africa to divert rivers to obtain alluvial diamonds. A channel is cut from one bend to the other and the river is dammed at both ends of the 'oxbow'. The dammed area is pumped dry and the gravel is excavated and examined.
Diamonds are a brittle substance despite it's hardness. It can shatter if struck by a sharp blow. In nature, diamond minerals need to be tough enough to survive in and gravel deposits. The minerals that are not tough enough to survive the continual shocks simply fracture, corrode away. Diamonds survive extremely well in this natural process.
Dredging - Large scale suction dredger and all material is examined for diamonds. Waste is discarded from the dredger into an area which already has been cleared.
Dredging is an excavation activity or operation usually carried out at least partly underwater, in shallow seas or fresh water areas with the purpose of gathering up bottom sediments and disposing of them at a different location. This technique is often used to keep waterways navigable.
Diverting Rivers - It is quite common in Africa to divert rivers to obtain alluvial diamonds. A channel is cut from one bend to the other and the river is dammed at both ends of the 'oxbow'. The dammed area is pumped dry and the gravel is excavated and examined.
Underground (Chambering, Block Caving & Sub-level Caving)
Chambering and Block caving are two underground mining techniques for extracting diamonds.
Sub-level caving uses a combination of both the chambering and block caving methods.
Sub-level caving uses a combination of both the chambering and block caving methods.
Primary and Secondary Deposits
Primary deposits are mined either by open-cast (open-pit) mines or underground mining.
Secondary mining is alluvial, coastal, or marine mining.
Secondary diamond deposits are concentrations of diamonds removed from primary deposits by natural processes at the Earth's surface. Secondary deposits can occur in the vicinity of the parent rock or any distance up to many hundreds of kilometers away. They can be found in ancient or modern river beds, beaches, or as sea-bed deposits.
Secondary mining is alluvial, coastal, or marine mining.
Secondary diamond deposits are concentrations of diamonds removed from primary deposits by natural processes at the Earth's surface. Secondary deposits can occur in the vicinity of the parent rock or any distance up to many hundreds of kilometers away. They can be found in ancient or modern river beds, beaches, or as sea-bed deposits.
Gravel is alluvial deposits of silt, sand, and gravel. They may not contain economic minerals such as diamonds.
Alluvial - made up of or found in the materials that are left by the water of rivers, floods, etc. Alluvium (from the Latin, alluvius, from alluere, "to wash against") is loose, unconsolidated (not cemented together into a solid rock) soil or sediments, which has been eroded, reshaped by water in some form, and redeposited in a non-marine setting. Alluvium is typically made up of a variety of materials, including fine particles of silt and clay and larger particles of sand and gravel.
Diamond is also found in rivers and streams or rocks around extinct volcanoes. This is because the igneous rock that makes up the volcano gets worn away by wind and rain over the years. Pieces of the igneous rock and diamond are carried down by the wind or in streams and rivers and, over many years, can get stuck together with lots of other pieces of sand and clay to form sedimentary rocks.
Surface Mining
Surface mining, including strip mining, open-pit mining andmountaintop removal mining, is a broad category of mining in which soil and rock overlying the mineral deposit (the overburden) are removed. It is the opposite of underground mining, in which the overlying rock is left in place, and the mineral removed through shafts or tunnels.
Open-Pit Mining
Open-pit mining, open-cut mining or opencast mining is a surface mining technique of extracting rock or minerals from the earth by their removal from an open pit or borrow.
This form of mining differs from extractive methods that require tunneling into the earth such as long wall mining. Open-pit mines are used when deposits of commercially useful minerals or rock are found near the surface; that is, where the overburden (surface material covering the valuable deposit) is relatively thin or the material of interest is structurally unsuitable for tunneling (as would be the case for sand, cinder, and gravel). For minerals that occur deep below the surface - where the overburden is thick or the mineral occurs as veins in hard rock - underground mining methods extract the valued material.
Hardness
Hardness is the ability to resist abrasion when a pointed fragment of another substance is drawn across it without sufficient pressure to develop fracture or cleavage.
The basis of hardness lies in the nature of and strength of the bonding between atoms, and not that the individual atoms of some element is stronger, tougher or more abrasive than others. Carbon atoms can combine in variety of ways.
The basis of hardness lies in the nature of and strength of the bonding between atoms, and not that the individual atoms of some element is stronger, tougher or more abrasive than others. Carbon atoms can combine in variety of ways.
Indicator Minerals
Indicator minerals are mineral species that, when appearing as transported grains in clastic sediments, indicate the presence in bedrock of a specific type of mineralization, hydrothermal alteration or lithology.
Indicator minerals are mineral species that, when appearing as transported grains in clastic sediments, indicate the presence in bedrock of a specific type of mineralization, hydrothermal alteration or lithology. Their physical and chemical characteristics, including a relatively high density, facilitate their preservation and identification and allow them to be readily recovered at the parts per billion level from sample media such as till, stream sediments or soil producing large exploration targets.
Indicator minerals for diamonds are spinel, diopside, and garnet.
Indicator minerals are mineral species that, when appearing as transported grains in clastic sediments, indicate the presence in bedrock of a specific type of mineralization, hydrothermal alteration or lithology. Their physical and chemical characteristics, including a relatively high density, facilitate their preservation and identification and allow them to be readily recovered at the parts per billion level from sample media such as till, stream sediments or soil producing large exploration targets.
Indicator minerals for diamonds are spinel, diopside, and garnet.
Resource, Reserve, Ore, Grade, Volume and Value
World Diamond Resource is summarized as all the diamond in the crust of the earth with current or future value. These include known deposits and yet to be found deposits.
Current Diamond Reserve is a stockpile in the ground which is geologically suitable for investment mining.
A Diamond Ore is a mineral deposit, such as a kimberlite occurrence, which is currently considered to be economically worth mining the economic content such as a diamond.
The Grade of the deposit assessed is the weight of the diamonds contained within a given quantity of ore deposit. The number of carats per tones of ore; that is the deposit rock such as gravel or kimberlite.
The Volume in terms of the diamond deposit is the quantity in terms of weight.
The Value of the diamond is the economic worth of the stones. The diamonds should be of high value, whatever the grade of the deposit. A low-grade deposit with large, high value stones may make the site worth developing.
Current Diamond Reserve is a stockpile in the ground which is geologically suitable for investment mining.
A Diamond Ore is a mineral deposit, such as a kimberlite occurrence, which is currently considered to be economically worth mining the economic content such as a diamond.
The Grade of the deposit assessed is the weight of the diamonds contained within a given quantity of ore deposit. The number of carats per tones of ore; that is the deposit rock such as gravel or kimberlite.
The Volume in terms of the diamond deposit is the quantity in terms of weight.
The Value of the diamond is the economic worth of the stones. The diamonds should be of high value, whatever the grade of the deposit. A low-grade deposit with large, high value stones may make the site worth developing.
Wednesday, April 16, 2014
Explain the covalent bonding
A chemical bond is a link between neighboring atoms caused by the interaction of electrons.
Electrons and Electronic forces are the key to all chemical activity. Chemistry is essentially, the action and reaction of electrons between atoms.
Covalent bonding is the sharing of electrons. Diamonds crystallize and covalent bindings occur.
Diamonds bond covalently and involve no attraction of opposite charges.
Ionic bonding occurs when atoms gain or lose electrons, the resulting electrically charged atoms are called ions.
Electrons and Electronic forces are the key to all chemical activity. Chemistry is essentially, the action and reaction of electrons between atoms.
Covalent bonding is the sharing of electrons. Diamonds crystallize and covalent bindings occur.
Diamonds bond covalently and involve no attraction of opposite charges.
Ionic bonding occurs when atoms gain or lose electrons, the resulting electrically charged atoms are called ions.
Overburden
Overburden is the material that lies above an area of economic interest. Overburden is distinct from tailings, the material that remains after economically valuable components have been extracted from the generally finely milled ore.
Overburden is removed during surface mining, but is typically not contaminated with toxic components and may be used to restore an exhausted mining site to a semblance of its appearance before mining began. Overburden may also be used as a term to describe all soil and ancillary material above the bedrock horizon in a given area.
A related term is interburden, meaning material that lies between two areas of economic interest, such as the material separating coal seams within strata.
The depth and composition of any overburden must also determine to calculate its effect on the economic viability.
Overburden is removed during surface mining, but is typically not contaminated with toxic components and may be used to restore an exhausted mining site to a semblance of its appearance before mining began. Overburden may also be used as a term to describe all soil and ancillary material above the bedrock horizon in a given area.
A related term is interburden, meaning material that lies between two areas of economic interest, such as the material separating coal seams within strata.
The depth and composition of any overburden must also determine to calculate its effect on the economic viability.
Origin, Occurrence and Locality
It's is importance to distinguish between the terms, origin, occurrence and locality. The origin are from the mantle of the earth, the occurrence are from kimberlite pipes and ancient beach beds, the locality would be Canada, Namibia, Botswana, etc.
Tuesday, April 15, 2014
Plastic Deformation
Plastic Deformation - dislocation of the crystal structure and occurs when diamond is deep within the earth. How hot it is and how much pressure. Tends to be more deep underground.
Why does this happen? Diamonds are brittle at room temperature but the atoms are mobile at extremely high temperatures. Dislocation and slipping occur along the glide plane.
Plastic is stated with dislocation is permanent. This concept is derived from geology. It distorts under pressure and does not return to its original shape after the pressure is released.
Causes brown color in many diamonds.
permanent deformation or change in shape of a solid body without fracture under the action of a sustained force. Small changes in the density of the crystals. It’s the distortion of the crystal growth and atomic structure the crystalline and pressure process.
There is elastic deformation, plastic deformation and brittle deformation.
This creates free bonds which may react to light to cause absorption. More light is absorbed toward the blue end of the spectrum, which results in brown color that is not dependent on the presence of nitrogen, or any other impurities which would cause color.
Why does this happen? Diamonds are brittle at room temperature but the atoms are mobile at extremely high temperatures. Dislocation and slipping occur along the glide plane.
Plastic is stated with dislocation is permanent. This concept is derived from geology. It distorts under pressure and does not return to its original shape after the pressure is released.
Causes brown color in many diamonds.
permanent deformation or change in shape of a solid body without fracture under the action of a sustained force. Small changes in the density of the crystals. It’s the distortion of the crystal growth and atomic structure the crystalline and pressure process.
There is elastic deformation, plastic deformation and brittle deformation.
This creates free bonds which may react to light to cause absorption. More light is absorbed toward the blue end of the spectrum, which results in brown color that is not dependent on the presence of nitrogen, or any other impurities which would cause color.
Subscribe to:
Posts (Atom)
